The
Origin of Life on Earth—Life from Nonlife
Geologists
estimate from radioisotope dating that the earth is approximately 4.6 billion
years old. The earliest direct evidence of life on earth are microfossils of 3.5 billion-year-old rock formations known as stromatolites from
In
trying to explain how life may have arisen, scientists first needed to know the
conditions that existed on the earth after its formation. In 1929, J. B. S. Haldane described the
atmosphere of primordial earth as a reducing atmosphere (with little free
oxygen present) containing primarily hydrogen, water, ammonia and methane. In 1953, Stanley Miller and Harold Urey constructed a reaction vessel in which they
duplicated the atmosphere Haldane described.
They heated the mixture to 80oC and provided the atmosphere
with an electrical spark to simulate lightning.
Over the course of a week, they removed samples from their system and
found a variety of common amino acids and other organic acids.
The
previous scenario for the origin of the first organic compounds, although once
widely accepted, is under increasing scrutiny.
Recent evidence suggests that carbon dioxide and nitrogen gas, not
methane and ammonia, were the major components of the earth’s primitive
atmosphere. These conditions are much
less favorable for the formation of organic compounds using the Miller/Urey
apparatus.
Scientists
have begun to look for new explanations of the origin of the first organic
chemicals and at older explanations, which are being revived. One of these is that life may have begun deep
in the oceans, in underwater
Another
explanation for the origin of the earth’s first organic molecules is that they
came from outer space. Astronomers are
detecting an increasing diversity of organic compounds (such as amino acids and
other hydrocarbons) in meteorites that have collided with the earth. Investigations of the most recent pass-by of
Halley’s Comet revealed that comets might be relatively rich in organic
compounds. Even though many scientists
think that the first organic compounds could have come from space, no microbial
life forms have been detected in space, and conditions in outer space are
incompatible with life as we know it.
A
second step in the origin of life must have been the hooking together of early
organic molecules into the polymers of living organisms: polypeptides,
polynucleotides and carbohydrates.
Organic molecules may have become isolated in tide pools or freshwater
ponds, and as water was lost through evaporation, condensation reactions could
have occurred. Alternatively, reacting
molecules may have been concentrated by adsorption on the surfaces of clay or
iron pyrite particles, where polymerization could occur.
The
final steps in the origin of life are the subject of endless speculation. In some way, organic molecules were surrounded
by a membrane-like structure, self-replication occurred and DNA became established
as the genetic material. A “chicken-or-egg”
paradox emerges if DNA was the first genetic material.
DNA codes for proteins, yet proteins (enzymes) are required for DNA
replication, transcription and translation.
Thomas R. Cech from the
What were the first living cell like? No one knows for sure because we don't have good data from the first 2 billion years of Earth's history—a period known as the Archaean. Most likely they were heterotrophic microbes that found enough energy from the acid and base molecules in the rich broth of the early seas to continue living and reproducing. These organisms extracted energy by fermentation, which is the same process used by unicellular microbes (mostly yeast) to make beer or bread.
As Earth cooled, geologic and atmospheric activity declined, and as gases thinkened the air, smaller amounts of solar radiation reached the surface. Lab experiments show that these changing conditions would have caused the organic soup to gradually thin, which is why we don't see a thick film of nutrients floating on today's oceans and rivers. The 'juicy' organic molecules on which heterotrophs could feed became more scarce, creating a crisis for the multiplying cells. The organic production of acids and bases via lightning, volcanoes, or solar radiation simply couldn’t satisfy the voracious appetite of the growing population of heterotrophs.
If life were to continue, another
source of nutrients was needed.
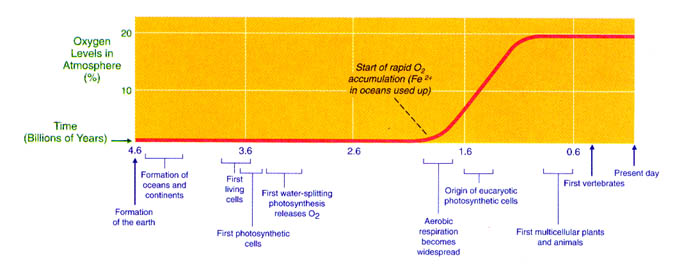
Photosynthesis, which is the production of organic molecules using solar
energy and inorganic compounds, solved the problem of a dwindling supply of
nutrients. The first photosynthetic
organisms probably used hydrogen sulfide as a source of hydrogen for reducing
carbon dioxide to sugar. Later, water
served this same purpose, and oxygen liberated by photosynthetic reactions
began to accumulate in the atmosphere.
Earth and its atmosphere slowly began to change. Ozone in the upper atmosphere began to filter
ultraviolet radiation from the sun, the reducing atmosphere slowly became an
oxidizing atmosphere, and at least some living organisms began to utilize
oxygen. About 420 million years ago
enough protective ozone had built up to make life on land possible. Ironically, the change from a reducing
atmosphere to an oxidizing atmosphere also meant that life could no longer
arise abiotically.